The ATM protein:
The protein produced by the Ataxia Telangiectasia Mutated gene is comprised of 3056 amino acids and has a weight of 370 kDa. ATM is involved in the repair pathway responsible for fixing DNA double stranded breaks. These types of damage are normal within a cell and are repaired through either homologous recombination (HR), which is important in the S and G2 stages of the cell cycle, or nonhomologous end joining (NHEJ), which is important for developing lymphocytes [1].
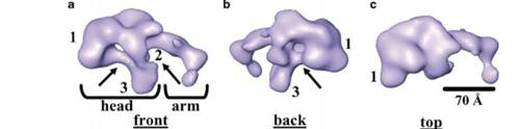
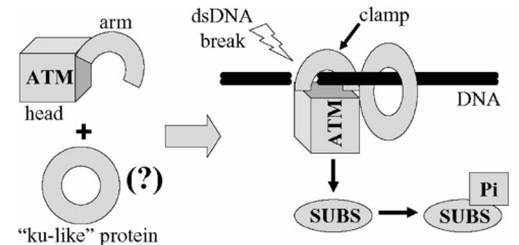
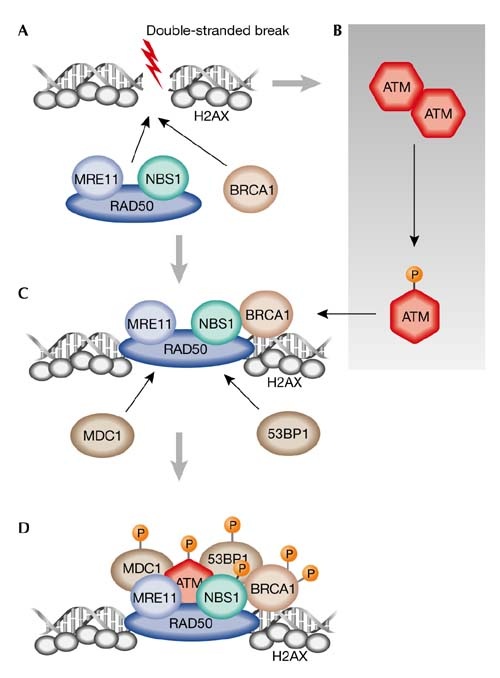
A double stranded break within the cell’s DNA triggers the activation of the ATM protein. ATM, which is present in its inactive multimeric form, becomes active when it is phosphorylated at Ser-1981, causing it to dissociate into its active monomeric state. This active ATM has been shown to interact with DNA through a structural “arm” domain present on the ATM protein (figures 1 and 2) [2]. From here ATM can act on other proteins which all work together to resolve the DNA double stranded break. In addition to its role in facilitating DNA repair, ATM also works to control cell-cycle checkpoints which are used to stop the cell from dividing if there is some sort of stress present (i.e. DNA damage) [3]. ATM’s role in the double stranded DNA damage response is represented in figure 3.
A double stranded break within the cell’s DNA triggers the activation of the ATM protein. ATM, which is present in its inactive multimeric form, becomes active when it is phosphorylated at Ser-1981, causing it to dissociate into its active monomeric state. This active ATM has been shown to interact with DNA through a structural “arm” domain present on the ATM protein (figures 1 and 2) [2]. From here ATM can act on other proteins which all work together to resolve the DNA double stranded break. In addition to its role in facilitating DNA repair, ATM also works to control cell-cycle checkpoints which are used to stop the cell from dividing if there is some sort of stress present (i.e. DNA damage) [3]. ATM’s role in the double stranded DNA damage response is represented in figure 3.
Figure 1: 3D structure of ATM using single-particle electron microscopy. All there structures are the same but are just rotated differently. [2]
Figure 2: ATM interaction with DNA upon the occurrence of a double stranded break. The ku-like protein is shown to depict the hypothesis that there might be other proteins, which interact with DNA in the same ring-like way, might bind with ATM. The SUBS stands for substrates which refers to the proteins ATM phosphorylates after it has become active. SUB with a Pi attached signifies the phosphorylated protein [2]. These various substrates are depicted in figure 3.
Figure 3: ATM and the DNA double stranded break repair pathway. The orange P being attached to the proteins signifies phosphorylation. This figure was taken from [3] and a more detailed explanation can be found by clicking on the figure.
References:
1. Yamamoto et al. (2012). Kinase-dead ATM protein causes genomic instability and early embryonic lethality in mice. Journal of Cell Biology, 198(3), 305-313. doi:10.1083/jcb.201204098
2. Llorca, O., Rivera-Calzada, A., Grantham, J., & Willison, K. (2003). Electron microscopy and 3D reconstructions reveal that human ATM kinase uses an arm-like domain to clamp around double-stranded DNA. Oncogene, 22(25), 3867-3874. doi:10.1038/sj.onc.1206649
3. McKinnon, P. (2004). ATM and ataxia telangiectasia. EMBO reprots, 5(8), 772-776. doi:10.1038/sj.embor.7400210
2. Llorca, O., Rivera-Calzada, A., Grantham, J., & Willison, K. (2003). Electron microscopy and 3D reconstructions reveal that human ATM kinase uses an arm-like domain to clamp around double-stranded DNA. Oncogene, 22(25), 3867-3874. doi:10.1038/sj.onc.1206649
3. McKinnon, P. (2004). ATM and ataxia telangiectasia. EMBO reprots, 5(8), 772-776. doi:10.1038/sj.embor.7400210
This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison