What is CLL?
|
Chronic Lymphocytic Leukemia (which can also be referred to as Small lymphocytic leukemia) is a cancer of the blood that is characterized by the accumulation of mature, incompetent B-Cells (lymphocytes). This disease is more common among the elderly population but there are a good amount of cases (about 1/3 of them) that are reported in patients who are under the age of 60 [1]. When looking at incidence rates around the globe it is found that there are more cases reported in North America and Europe as compared to Africa and Asia where CLL is not as common [2].
The diagnosis of CLL, which can be caught by doing a routine blood test, usually comes as a surprise to most as a majority of the patients diagnosed are asymptomatic at the time. As the disease progresses symptoms such as fatigue, shortness of breath, night sweats, and swollen lymph nodes/spleen can develop [3]. The reason that CLL is a problem is that the B-cells that are normally supposed to undergo apoptosis (programed cell death) are not due to intracellular abnormalities that result in the cell ignoring the death signal. These death resistant immune cells travel through the body and can accumulate in places such as the spleen, bone marrow, lymph nodes as well as other organs where they can form tumors [3]. Other risks that are concurrent with CLL are recurrent infections as well as the development of hemolytic anemia which is a type of auto immune disease that results in the premature breakdown of red blood cells [2]. |
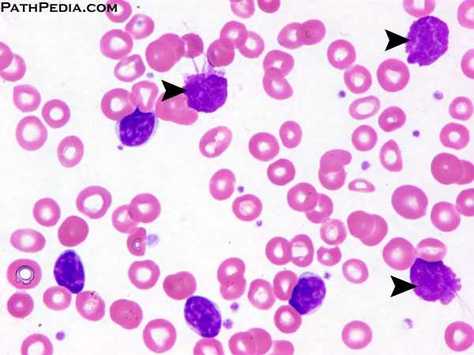
Figure 1: The arrows indicate typical morphology of CLL cells.
|
Treatment of CLL
Treatment of patients with Chronic Lymphocytic Leukemia tends to be variable and is dependent upon four types of prognostic markers present upon diagnosis. These four markers are physical features such as age and sex, disease activity which can be monitored by looking at factors such as lymphocyte count and bone marrow infiltration pattern, serum markers which look at the types of markers on the surface of the cancer cells , and genetic markers such as mutations in ATM and Ig heavy chain variable regions (VH regions) [1]. Along with these factors, doctors also take into account whether the patient is experiencing symptoms at the time of diagnosis. Based on the types of markers present and the presence/absence of symptoms, doctors can better determine whether it is better to start treatment or to actively monitor the patient and delay treatment until the disease progresses. The Rai and Binet staging system is frequently used by doctors to better assess the status of the patient [4]. Once the doctor and patient determine that it is time for treatment there are a handful of drugs that are available. The type of chemotherapy drugs include alkylating agents such as bendamustine (better known as Treanda), and purine analogs such as fludarabine. Monoclonal antibodies are another type of drug that is used that works by targeting certain markers on the leukemic cells and destroys them. There are a variety of combinations of these drugs that are used when patients in order to maximize effectiveness. For those patients with a more aggressive disease, a bone marrow transplant is possible but this procedure is more risky and requires an exact match in order for the patient’s body to accept the marrow as its own [3]. Though CLL is considered to be an incurable disease, some patients do go into remission and live out the rest of their lives without the reoccurrence of CLL.
Figure 2: Markers of B-lymphocytes: The above figure shows potential markers one would find on a B-Cell. When diagnosing CLL tests are done to look for the presence of CD5, CD19, CD20, and CD23 [8]
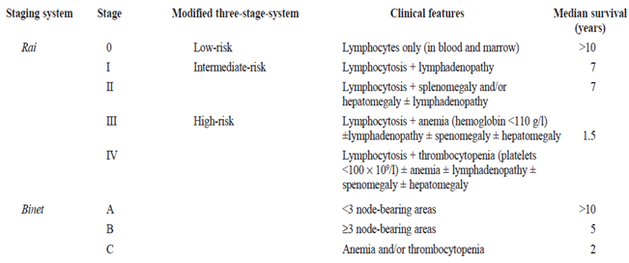
Figure 3: The Rai and Binet staging system: This table shows the classifications used to determine what stage a patient is at. [4]
A Genetic Component: Ataxia-Telangiectasia Mutated and CLL
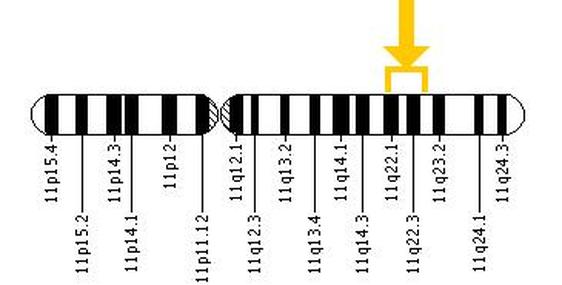
There are many genetic abnormalities that can are observed when studying CLL B-Cells from various patients. Of these abnormalities, a deletion of the q arm (long arm) of chromosome 11, denoted as an 11q-, is present in about 20% of CLL cases. This finding brought attention to Ataxia-Telangiectasia Mutated, better known as ATM, which is located within the 11q22-23 region [5] (Figure 4).
Figure 4: Here is a map of chromosome 11. The yellow arrow indicates the location of the ATM gene.
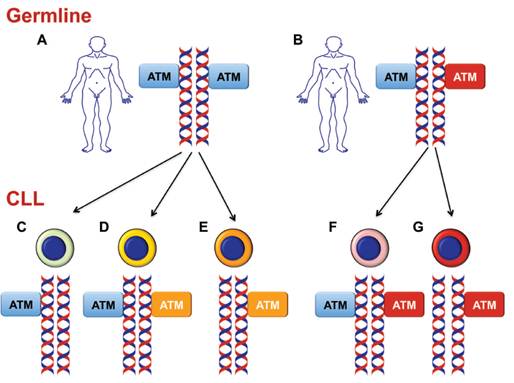
Patients with this chromosomal abnormality are said to have a poorer outcome, having higher odds of experiencing chemorefractoriness (failure to respond to chemotherapy) [4]. Though the disruption of ATM is caused by deletions in a majority of cases, mutations at the ATM locus have also been found in some cases. These disruptions at the ATM locus can either be acquired sporadically in somatic cells or a mutation could be present in the patient’s germline (Figure 5). Interestingly enough, it has been shown that these germline mutations have a more substantial effect on the progression of CLL rather than the initiation of the disease [6].
Figure 5: This figure illustrates how ATM mutations can be acquired in CLL cells. The blue boxes indicate normal ATM alleles while the red indicate ATM mutations within the germline. Points C, E, and G illustrate deletions of the ATM gene shown by the fact that only one ATM box is present. Yellow boxes indicate somatic mutations of the ATM gene. This figure is from [9].
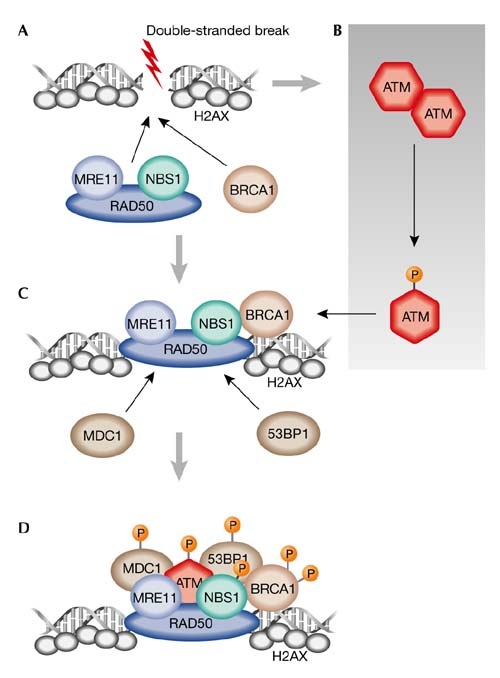
The reason this gene is of interest to researchers is because ATM is an important gene that plays a role in regulating the DNA damage response of cells. When DNA damage is detected within a cell this response is activated which temporally halts the cell cycle while the DNA damage is repaired, or the cell is killed via apoptosis if the damage is too great [5]. Ataxia-Telangiectasia Mutated is known to be one of the bigger proteins, having 66 exons that encode a protein of about 370 kDa. The ATM protein belongs to the phosphatidylinostidylinositol-3-OH kinase family (PI(3)K family) and is at the pinnacle of the DNA damage response pathway. It is a nuclear serine-threonine kinase that undergoes autophosphorylation at Ser-1981 upon activation, causing it to dissociate into its active monomeric state where it then acts downstream by inducing other proteins in the DNA damage pathway such as H2AX, 53BP1, MDC1 and BRCA1 which work to enforce cell cycle checkpoints, preventing further division until the damage is repaired [7].
Figure 6: ATM and the DNA double stranded break repair pathway. The orange P being attached to the proteins signifies phosphorylation. This figure was taken from [7] and a more detailed explanation can be found by clicking on the figure.
References:
Header Image: http://extremebodyfit.com/muscle-building/genetics-and-muscle-growth
1. Byrd, J., Stilgenbauer, S., & Flinn, I. W. (2004). Chronic Lymphocytic Leukemia. American Society of Hematology, 2004(1), 163-183. doi:10.1182/asheducation- 2004.1.163
2. Zenz, T., Mertens, D., Kuppers, R., Dohner, H., & Stilgenbauer, S. (2010). From pathogenesis to treatment of chronic lymphocytic leukaemia. Nature Reviews Cancer, 10(1), 37-50. doi:10.1038/nrc2764
3. http://www.lymphoma.org/site/pp.asp?c=bkLTKaOQLmK8E&b=6300147
4. Kalil, N., & Cheson, B. (1999). Chronic Lymphocytic Leukemia. The Oncologist, 4(5), 352-369. Retrieved from http://theoncologist.alphamedpress.org/content/4/5/352.short
5. Guarini et. al. (2012). ATM gene alterations in chronic lymphocytic leukemia patients induce a distinct gene expression profile and predict disease progression. Haematologica, 97(1), 47-55. doi:10.3324/haematol.2011.049270
6. Skowronska, A., Austen, B., Powell, J. E., Weston, V., Oscier, D. G., Dyer, M. J., . . . Stankovic, T. (2012). ATM germline heterozygosity does not play a role in chronic lymphocytic. Haematologica, 97(1), 142-146. doi:10.3324/haematol.2011.048827
7. McKinnon, P. (2004). ATM and ataxia telangiectasia. EMBO reprots, 5(8), 772-776. doi:10.1038/sj.embor.7400210
8. http://archive.constantcontact.com/fs161/1104839880793/archive/1111245176003.html
9. Rossi, D., & Gaidano, G. (2012). ATM and chronic lymphocytic leukemia: mutations, and not only deletions, matter. Haematologica, 97(1), 5-8. doi:10.3324/haematol.2011.057109
1. Byrd, J., Stilgenbauer, S., & Flinn, I. W. (2004). Chronic Lymphocytic Leukemia. American Society of Hematology, 2004(1), 163-183. doi:10.1182/asheducation- 2004.1.163
2. Zenz, T., Mertens, D., Kuppers, R., Dohner, H., & Stilgenbauer, S. (2010). From pathogenesis to treatment of chronic lymphocytic leukaemia. Nature Reviews Cancer, 10(1), 37-50. doi:10.1038/nrc2764
3. http://www.lymphoma.org/site/pp.asp?c=bkLTKaOQLmK8E&b=6300147
4. Kalil, N., & Cheson, B. (1999). Chronic Lymphocytic Leukemia. The Oncologist, 4(5), 352-369. Retrieved from http://theoncologist.alphamedpress.org/content/4/5/352.short
5. Guarini et. al. (2012). ATM gene alterations in chronic lymphocytic leukemia patients induce a distinct gene expression profile and predict disease progression. Haematologica, 97(1), 47-55. doi:10.3324/haematol.2011.049270
6. Skowronska, A., Austen, B., Powell, J. E., Weston, V., Oscier, D. G., Dyer, M. J., . . . Stankovic, T. (2012). ATM germline heterozygosity does not play a role in chronic lymphocytic. Haematologica, 97(1), 142-146. doi:10.3324/haematol.2011.048827
7. McKinnon, P. (2004). ATM and ataxia telangiectasia. EMBO reprots, 5(8), 772-776. doi:10.1038/sj.embor.7400210
8. http://archive.constantcontact.com/fs161/1104839880793/archive/1111245176003.html
9. Rossi, D., & Gaidano, G. (2012). ATM and chronic lymphocytic leukemia: mutations, and not only deletions, matter. Haematologica, 97(1), 5-8. doi:10.3324/haematol.2011.057109
This web page was produced as an assignment for Genetics 677, an undergraduate course at UW-Madison